Introduction

Nature v Nurture – what is epigenetics?
It’s a commonly asked question, especially in the last 30 years, because epigenetics is a young area of literature. Have you heard of the concept “nature vs. nurture?” Well, that is the study of epigenetics.
To understand epigenetics, you first have to understand what genetics is. Genetics is the “nature” component of epigenetics.
Genetics is the study of genes, but what are genes? Genes are segments of DNA that code for certain things in our bodies – it’s like a powerful “master recipe book” that makes you…well, you! Whether the DNA is coding for an amino acid, a protein, a hormone, or a network of structures in your cells that influence cellular functions…even your behaviour as a whole human being is influenced by DNA.
Here’s the kicker: EVERY cell in your body has the exact same DNA. A brain cell has the same DNA as a muscle cell, a skin cell, a stomach cell or a liver cell (or ANY type of cell). Every cell in your body has a copy of the “master recipe book”. However, particular genes are silenced, which allows us to have different cell types. The cells in your brain will not express stomach cell DNA, and your liver cells won’t activate the muscle cell DNA.
So, how come all cells in your body have the exact same DNA, but you have thousands of different cell types? Well, reader, meet “nurture”. Nurture is a fancy way of saying environment. Even when you were a teeny tiny clump of cells in your mother’s womb, nurture was playing a part in your development. Those cells all started out looking the same and all having the potential to be ALL types of cells. But then, as the cells grew, the environment started to vary at different sites of the cells. DNA started to get expressed in some parts, while in other parts, it would shut off. This is called “differentiation”, which takes us from a clump of identical cells to a fully functioning body with an orchestra of different systems and organs carrying out their unique functions.
But then, that’s not epigenetics! That’s differentiation! Ahh, well, the process of epigenetics is quite similar; they are the two sides of the same coin (1). DNA is there in our cells. Some of it is expressed, some of it is inhibited. The difference about epigenetics is that “epi” means “above”; it doesn’t just happen in our bodies. We pass them on to our children. Are you ready to sink your teeth in?
How does epigenetics work?
Let’s start with an analogy about smartphones since everyone has a phone. DNA code (genes) is like your phone’s hard coding and programming. Android or iOS phones are what they are because of how they’ve been developed.
Epigenetics is like going into your phone and changing the settings. The hard wiring isn’t affected, but the phone’s functions are changed. But to take the analogy further, the thing that makes it epigenetics is that these changes to the settings have been saved and turned into “factory settings” and then passed down through generations, aka epigenetic inheritance. Anyone can change their settings (epigenetics) with the right environment but can’t change the original manufacturing code (DNA code).
Don’t like phones? Okay, how about TV or radio for the following analogy?
DNA codes (genes) are like lots and lots of channels. They’re all there, and they’re all broadcasting. However, you can choose which channels to turn the volume up on, which to silence, and which to tune into (epigenetics). The channels you tune in to get passed down through generations, which is also known as epigenetic inheritance. However, individuals can always change what they’ve inherited by choosing which channels they want to tune into.
So we’ve talked about analogies, but what about the actual nanocellular process that’s going on? Your DNA is a series of very, very long, continuous strands of purines and pyrimidines (these are the basic building blocks of your genes). Patches of these codes are different genes, each with a location in the strand. The thing is, DNA isn’t just sitting all tangled up in your cells. It’s structured ingeniously. It’s tightly coiled around histones, which are protein structures. Imagine hair (DNA) twisted and coiled around hair curlers (histones). If it keeps uncoiling, the strand of DNA can be accessed by cells, and cellular functions can start being instructed.
The biochemical process of epigenetic modification
This can happen in 2 main ways (1):
1. Histone modification is a very structural process. Imagine turning a tap in the kitchen sink, you loosen it, and it’s on. You tighten it, and it’s off.
Acetylation (a chemical process) can switch genes ON because it modifies the structure of the histones (hair curlers) to loosen up, making the genetic code accessible and active!
Methylation (a different chemical process) can switch genes OFF because its reaction modifies the structure of histones and makes them so much tighter, making the genetic code Inaccessible and inactive!
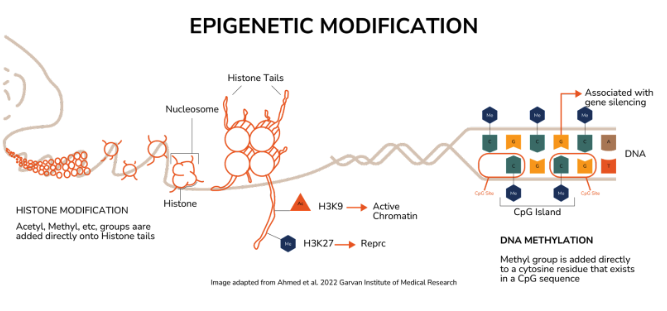
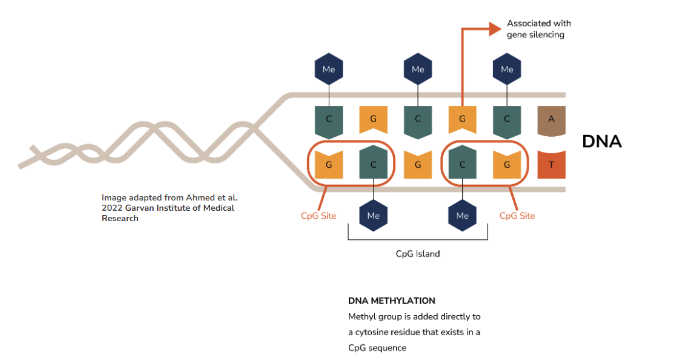
2. DNA methylation is a chemical process *right* at the site of the DNA strand. Imagine it’s like a toggle on and off a light switch.
The bases of the DNA (purines and pyrimidines), but more specifically a stretch of DNA full of cytosine bases repeating after each other (it’s a stretch of CCCCCCCCCC), can undergo a chemical process where a methyl group can be added directly to each of those Cs. A stretch of Cs that is all methylated is a form of gene silencing, like turning the light off, and this methylation process is reversible.
So, there are multiple ways to reversibly turn genes on or off at different sites. These changes are inherited, but they can be modified.
That’s great news, but that brings us to the next question!
WHAT in our environment can cause epigenetic modification?
(In other words, how does your lifestyle influence the biochemical processes of “methylation” and “acetylation” that change how your genes work?
One of our favourite studies was with mother rats and their little pups. Depressed mumma rats wouldn’t lick their babies so that they wouldn’t bond, and the pups exhibited signs and symptoms of depression as they grew. When the research team examined the brain DNA of these depressed mumma rats, they found a methylation around the gene responsible for licking behaviour, meaning the gene was turned off. These depressed pups that grew into mature rats also wouldn’t lick their babies, having inherited this epigenetic configuration.
On the other hand, while happy mumma rats would indeed lick their babies, the DNA in their brains did NOT have this methylation around the licking gene. Therefore, the mumma rats could bond with their babies, and those pups grew into happy mumma rats. The licking behaviour, along with the epigenetic configuration, passed on.
Here’s the exciting part: this study switched around the pups, giving depressed pups to happy mummas and happy pups to depressed mummas. The mummas in the respective groups licked or did not lick their babies.
The researchers found that depressed pups licked by happy mumma rats experienced a change in their brains—the methylation on that particular gene disappeared! Sadly, the happy rats given to depressed mumma rats were not licked, and their brains developed methylation around the licking gene (where it previously hadn’t been). Their behaviour also began to exhibit depression (2-6).
Although that was an animal study, there is now a lot of research and data on how epigenetics in humans play out (8)!
Environmental factors, such as pollutants, heavy metals, and radiation (9), cause epigenetic modification. Although we may not always have control over these particular environmental factors, being more intentional about our living situation and environment helps.
There are also lifestyle factors and behaviours – things you have control over – to influence epigenetic modification. Such factors include diet, drugs, and cigarettes9. There are a myriad of human twin studies (10) that have identified epigenetic differences in people who have gestational diabetes (11), type 2 diabetes (12), rheumatoid arthritis (13), obesity (14), depression (15 )and stroke (16).
So, how does Chiropractic care fit into epigenetic modification?
Many alternative medicine approaches have produced unexplainable outcomes or “miracles.” However, recent studies of epigenetics have begun to illuminate the effects of holistic medicines, like chiropractic care, scientifically helping to explain the innate intelligence that our cells have (17).
Remember our “Atomic Habits” blog, where we discussed the newly found connection between chiropractic care and reduction of major depressive disorder (18)? Epigenetic studies have also found that depression can be induced or reduced by epigenetic factors (8,15). It is now scientifically proven that chiropractic care facilitates neuroplasticity and changes in the brain (19,20,21). Neuroplasticity causing changes in the brain is an epigenetic process and occurrence in itself (16,22).
This field of chiropractic and epigenetic science is still young, as more studies are needed to investigate the relationship between chiropractic care and epigenetic modification specifically. However, we can draw the connections between chiropractic care, neuroplasticity, and epigenetics with the data that has recently come to the surface. There’s so much more coming into the world of research. Still, if your brain can change, that is an example of epigenetic modification, and that is powerful information.
So next time you’re offered the option of seeing a chiropractor, remember that it’s never just about pain. Instead, think about the deeper possibilities that it presents. Chiropractic care supports and retrains the nervous system, and it has been proven to cause beneficial neuroplastic changes to the brain. That is an excellent example of epigenetics!
References:
1. Heard E, Martienssen RA. Transgenerational epigenetic inheritance: myths and mechanisms. Cell. 2014 Mar 27;157(1):95-109. doi: 10.1016/j.cell.2014.02.045. PMID: 24679529; PMCID: PMC4020004. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4020004/
2. Liu D, Diorio J, Day JC, Francis DD, Meaney MJ. Maternal care, hippocampal synaptogenesis and cognitive development in rats. Nat Neurosci. 2000;3:799–806.
3. Weaver IC, Cervoni N, Champagne FA, et al. Epigenetic programming by maternal behavior. Nat Neurosci. 2004;7:847–54.
4. Weaver IC, Meaney MJ, Szyf M. Maternal care effects on the hippocampal transcriptome and anxiety-mediated behaviors in the offspring that are reversible in adulthood. Proc Natl Acad Sci USA. 2006;103:3480–5.
5. Weaver IC, D’Alessio AC, Brown SE, et al. The transcription factor nerve growth factor-inducible protein a mediates epigenetic programming: altering epigenetic marks by immediate-early genes. J Neurosci. 2007;27:1756–68.
6. Meaney MJ, Szyf M. Environmental programming of stress responses through DNA methylation: life at the interface between a dynamic environment and a fixed genome. Dialogues Clin Neurosci. 2005;7:103–23.
7. Moosavi A, Motevalizadeh Ardekani A. Role of Epigenetics in Biology and Human Diseases. Iran Biomed J. 2016 Nov;20(5):246-58. doi: 10.22045/ibj.2016.01. Epub 2016 Jul 5. PMID: 27377127; PMCID: PMC5075137.
8. Szyf, M., Weaver, I.C., Champagne, F.A., Diorio, J. and Meaney, M.J., 2005. Maternal programming of steroid receptor expression and phenotype through DNA methylation in the rat. Frontiers in neuroendocrinology, 26(3-4), pp.139-162.
9. Metere A, Graves CE. Factors Influencing Epigenetic Mechanisms: Is There A Role for Bariatric Surgery? High Throughput. 2020 Mar 20;9(1):6. doi: 10.3390/ht9010006. PMID: 32244851; PMCID: PMC7151212. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7151212/
10. Bell, J.T. and Spector, T.D., 2011. A twin approach to unraveling epigenetics. Trends in Genetics, 27(3), pp.116-125.
11. Elliott, H.R., Sharp, G.C., Relton, C.L. et al. Epigenetics and gestational diabetes: a review of epigenetic epidemiology studies and their use to explore epigenetic mediation and improve prediction. Diabetologia 62, 2171–2178 (2019). https://doi.org/10.1007/s00125-019-05011-8
12. Ling, C., Bacos, K. & Rönn, T. Epigenetics of type 2 diabetes mellitus and weight change — a tool for precision medicine?. Nat Rev Endocrinol 18, 433–448 (2022). https://doi.org/10.1038/s41574-022-00671-w
13. Viatte, S., Plant, D. & Raychaudhuri, S. Genetics and epigenetics of rheumatoid arthritis. Nat Rev Rheumatol 9, 141–153 (2013). https://doi.org/10.1038/nrrheum.2012.237
14. Mahmoud AM. An Overview of Epigenetics in Obesity: The Role of Lifestyle and Therapeutic Interventions. Int J Mol Sci. 2022 Jan 25;23(3):1341. doi: 10.3390/ijms23031341. PMID: 35163268; PMCID: PMC8836029.
15. Signe Penner-Goeke & Elisabeth B. Binder (2019) Epigenetics and depression , Dialogues in Clinical Neuroscience, 21:4, 397-405, DOI: 10.31887/DCNS.2019.21.4/ebinder
16. Felling RJ, Song H. Epigenetic mechanisms of neuroplasticity and the implications for stroke recovery. Exp Neurol. 2015 Jun;268:37-45. doi: 10.1016/j.expneurol.2014.09.017. Epub 2014 Sep 26. PMID: 25263580; PMCID: PMC4375064.
17. Kanherkar RR, Stair SE, Bhatia-Dey N, Mills PJ, Chopra D, Csoka AB. Epigenetic Mechanisms of Integrative Medicine. Evid Based Complement Alternat Med. 2017;2017:4365429. doi: 10.1155/2017/4365429. Epub 2017 Feb 21. PMID: 28316635; PMCID: PMC5339524. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5339524/
18. Kiani AK, Maltese PE, Dautaj A, Paolacci S, Kurti D, Picotti PM, Bertelli M. Neurobiological basis of chiropractic manipulative treatment of the spine in the care of major depression. Acta Biomed. 2020 Nov 9;91(13-S):e2020006. doi: 10.23750/abm.v91i13-S.10536. PMID: 33170171; PMCID: PMC8023121.
19. Maltese PE, Michelini S, Baronio M, Bertelli M. Molecular foundations of chiropractic therapy. Acta Biomed. 2019 Sep 30;90(10-S):93-102. doi: 10.23750/abm.v90i10-S.8768. PMID: 31577263; PMCID: PMC7233649.
20. Lelic, D, Niazi, IK, Holt, K, Jochumsen, M, Dremstrup, K, Yielder, P, Murphy, B, Drewes, A and Haavik, H (2016), “Manipulation of dysfunctional spinal joints affects sensorimotor integration in the pre-frontal cortex: A brain source localization study,” Neural Plasticity, Volume 2016.
21. Haavik H, Niazi IK, Holt K, Murphy B. Effects of 12 Weeks of Chiropractic Care on Central Integration of Dual Somatosensory Input in Chronic Pain Patients: A Preliminary Study. J Manipulative Physiol Ther. 2017 Mar-Apr;40(3):127-138. doi: 10.1016/j.jmpt.2016.10.002. Epub 2017 Feb 10. PMID: 28196631.
22. Nayak, M.; Das, D.; Pradhan, J.; Ahmed, R.G.; Laureano-Melo, R.; Dandapat, J. Epigenetic signature in neural plasticity: The journey so far and journey ahead. Heliyon 2022, 8, e12292.

Recent Comments